 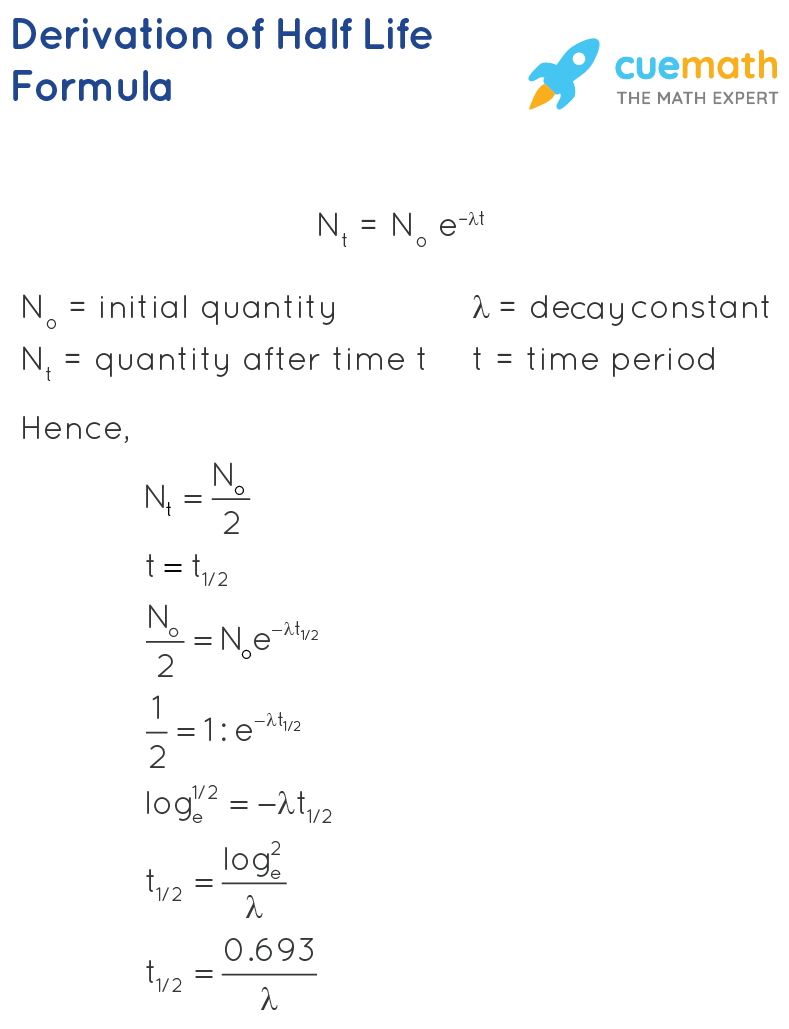
Some examples would be the Water Hyancith weed or the Japonese Honeysuckle 
Invasive species - These can be any living thing that is not native to a location and causes harm to the enviormant. Another example would be major pandemics, like covid 19. Wildfires - With the relation to the enviorment aroun it, we can calculate the exponential rate at which the fire will continue to spreadĭiseases - Cancer, cancer cells spread exponentially if not treated. This can help with calculating when food will start going bad to give products expiration rates. One of the most common uses of Exponential Growth is to calculate population growth, but there are still many other important uses such as:Ĭompound Interest - Compound Interest at a constant rate provides exponential growth to capital īiology - Studying the growth at which micro-organisms reproduce in a suitable evniorment. There are also other formulas used to calculate exponential growth shown below: When we look up Exponential Growth on the dictionary it defines it as "Growth of a system in which the amount being added to the system is proportional to the amount already present: the bigger the system is, the greater the increase."Īlthough there is no single person credited to the discovery or creation of Exponential growth and decay, we can still give credit to the English economist and scholar Thomas Malthus who came up with the formula while trying to prove that population growth was related to the economy in the late 1700's.Įven though this formula is still valid, we are more used to seeing this formula for calculating Exponential Growth: Let's take a look at Exponential growth first. There are two key words that really define exponential growth, those are proportional and constant. In simple terms Exponential growth is the increases of something at contstant rate proportional to it's size, the same applies for decay except the value would be decreasing at a constant rate. If a rock sample is crushed and the amount of Ar-40 gas that escapes is measured, determination of the Ar-40:K-40 ratio yields the age of the rock.Exponential growth and decay are formulas that are very commonly used in the real world to predict trends and changes of something. K-40 decays by positron emission and electron capture to form Ar-40 with a half-life of 1.25 billion years. Potassium-argon dating uses a similar method. If there is additional lead-206 present, which is indicated by the presence of other lead isotopes in the sample, it is necessary to make an adjustment. This assumes that all of the lead-206 present came from the decay of uranium-238. Therefore, by measuring and analyzing the ratio of U-238:Pb-206, we can determine the age of the rock. In a sample of rock that does not contain appreciable amounts of Pb-208, the most abundant isotope of lead, we can assume that lead was not present when the rock was formed. Since U-238 has a half-life of 4.5 billion years, it takes that amount of time for half of the original U-238 to decay into Pb-206. For example, uranium-238 (which decays in a series of steps into lead-206) can be used for establishing the age of rocks (and the approximate age of the oldest rocks on earth). Radioactive dating can also use other radioactive nuclides with longer half-lives to date older events. 
Radioactive Dating Using Nuclides Other than Carbon-14 Samples tested during and after this period must be checked against another method of dating (isotopic or tree rings). Fission bombs ignite to produce more C-14 artificially. Lastly, accuracy of C-14 dating has been affected by atmosphere nuclear weapons testing. For this reason, aquatic samples cannot be effectively C-14 dated. Substances must have obtained C-14 from the atmosphere. Before or after this range, there is too little of the isotope to be detected. For example, a sample can be C-14 dating if it is approximately 100 to 50,000 years old. (CC-BY 4.0 OpenStax).Ĭ-14 dating does have limitations. Comparing this ratio to the C-14:C-12 ratio in living organisms allows us to determine how long ago the organism lived (and died). After death, the C-14 decays and the C-14:C-12 ratio in the remains decreases. \): Along with stable carbon-12, radioactive carbon-14 is taken in by plants and animals, and remains at a constant level within them while they are alive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
